Cooling, chilling and cold stabilization in food industry
Back to EFFICIENCY FINDER
Back to EFFICENCY FINDER OF FOOD INDUSTRY
1. OBJECTIVE
Cooling is used to reduce the temperature of the food from one processing temperature to another or to a required storage temperature. Chilling is a processing technique in which the temperature of a food is reduced and kept in a temperature between -1 to 8°C. The objective of cooling and chilling is to reduce the rate of biochemical and microbiological changes in foods, to extend the shelf-life of fresh and processed food, or to maintain a certain temperature in a food process, e.g. in fermentation and treatment of beer. Cooling is also used to promote a change of state or aggregation, e.g. crystallization. The objective of cold stabilization is to precipitate our tartrates in wines, or fatty acids in spirits before bottling (BAT in the Food, Drink and Milk Industries, June 2005).
2. FIELD OF APPLICATION
Cooling, chilling and cold stabilization are widely used in the food industry sector. Chilling is used for preservation of a lot of perishable foods. In the wine sector, cooling and chilling are applied to clarify the must before fermentation. Cold stabilization is used in the beer, wine and spirit sectors. Beer is cold stabilized to precipitate the protein-polyphenol adduct. The beer is kept between -2 to -3°C for at least 12 hours (BAT in the Food, Drink and Milk Industries, June 2005).
3. DESCRIPTION OF TECHNIQUES, METHODS AND EQUIPMENT
- Cooling
- (BAT in the Food, Drink and Milk Industries, June 2005)
- Cooling of liquid foods is commonly carried out by passing the product through a heat exchanger or cooler or cooling the vessels. The cooling medium in the cooler can be ground water, water recirculating over a cooling tower, or water, eventually mixed with agents like glycol, which is recirculated via a mechanical refrigeration system or ice-water system. The equipment used for freezing can also be used for cooling.
- Chilling
- (BAT in the Food, Drink and Milk Industries, June 2005)
- The supply of chilled food to consumers requires a sophisticated contribution system, involving chilled stores, refrigerated transport and chilled retail display cabinets. Chilled food can be grouped into three categories according to the storage temperature. A fourth category is applied to winemaking:
- 1. -1 to 1°C for fresh fish, meat, sausages and ground meats, smoked meats and fish
- 2. 0 to 5°C for pasteurized canned meat, milk and milk products, prepared salads, baked goods, pizzas, unbaked dough and pastry
- 3. 0 to 8°C for fully cooked meat and fish pies, cookd or uncooked cured meats, butter, margarine, cheese and soft fruits
- 4. 8 to 12°C for wine industry. The must is kept at this temperature for 6 to 24 hours.
- The equipment used for freezing can also be used for chilling.
- Cold stabilization
- (BAT in the Food, Drink and Milk Industries, June 2005)
- Cold stabilization is a technique for chilling wines before bottling to cause the precipitation of tartrate crystals. Three techniques can be employed:
- 1. Cold stabilization by batch and stabulation:
- Cold stabilization by batch and stabulation is the oldest technique. It consists of bringing the wine to a temperature below zero close to freezing point and then stabulating it in an isothermal tank for a period of five to eight days.
- 2. Continuous cold stabilization:
- In continuous cold stabilization the stabulation tank is replaced by a cylindro-conical crystalliser and an agitator, in which the wine will remain for only 30 to 90 minutes.
- 3. Cold stabilization by crystal seeding:
- Cold stabilization is carried out by crystal seeding, consisting of refrigerating at between -1 and -2°C and seeding at 4g/l of tartaric crystals with agitation over 2 to 4 hours, and later storage in tanks, followed by decantation after 12 to 48 hours.
The two last techniques are the most widely-used. There can be many variations on these basic schemes.
4. COMPETITIVE TECHNOLOGIES AND ENERGY SAVING POTENTIALS
- a) Changes in the process
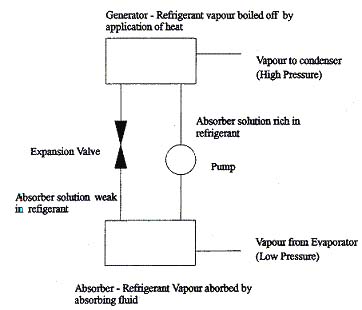
- Absorption cooling: (Galitsky et al. Improving Energy Efficiency in Pharmaceutical Manufacturing Operations -- Part II: HVAC, Boilers and Cogeneration, Lawrence Berkeley National Laboratory, publication number LBNL 60288 Part 2)
- Adsorption cooling utilizes the capacity of certain substances to adsorb water on their surface, from where it can be separated again with the application of heat. Adsorption cooling units can utilize waste heat from a variety of processes, where for standard refrigeration units, electricity would be required. These systems do not use ammonia or corrosive salts, but use silica gel (which also helps to reduce maintenance costs). Adsorption units were originally developed in Japan and are now also marketed in the United States.
Figure 1: Typical absorption cooling cycle (LITERATURE: http://lorien.ncl.ac.uk/ming/cleantech/refrigeration.htm)
- Cryogenic cooling: (BAT in the Food, Drink and Milk Industries, June 2005)
- Cryogenic cooling systems are open systems, in comparison to the conventional mechanic ones, that expel the spent vapour into the atmosphere without recompressing it.
- In cryogenic cooling, the food is in direct contact with the refrigerant, which may be solid or liquid carbon dioxide or liquid nitrogen. As the refrigerant evaporates or sublimates, it removes heat from the food, thereby causing rapid cooling. Both liquid carbon dioxide and nitrogen refrigerants are colourless, odourless and inert.
- Cooling in the cryogenic process can be achieved in short times. However, overall, the energy consumption in conventional cooling is significantly less than the total energy required for the manufacture and use of liquid nitrogen and carbon dioxide.
- Use of magnetocalorific materials:
(LITERATURE:
Goho, A. (2004) Cool magnet: a little bit of iron gives magnetic refrigeration a boost.
Engelbrecht, K. L., (2005) A Numerical Model of an Active Magnetic Regenerative Refrigeration System, Thesis, Mechanical Engineering, University of Wisconsin – Madison)
- Use of magnetocalorific materials:
- To make refrigerators and air conditioners more energy efficient, several research groups around the world are developing magnetic-refrigerant materials. According to Goho (2004), magnetic-cooling systems could also be less polluting than current systems because they wouldn't use environmentally harmful chemicals, such as ammonia or chlorofluorocarbons.
- When a magnetic-refrigerant material is exposed to a magnetic field, the field forces the spins of electrons in the material to align. As a result, the material heats up. Removing the field permits the electrons to relax into less-ordered states, and the material cools down. By cycling the material through these hot and cold states and venting away the heat, the system can generate an overall cooling effect (Goho 2004).
- Researchers at the Department of Energy's Ames (Iowa) Laboratory and the Astronautics Corporation of America in Madison, Wis., have created a prototype magnetic refrigerator that operates at room temperature (Weiss 2002).
- In 2005, studies by the University of Wisconsin predicted that magnetic refrigeration systems can operate more efficiency than current baseline vapor compression systems - if the regenerator is adequately large. According to Engelbrecht et al. (2005), the challenge now is to find more advanced magnetocaloric materials that are economical and allow design of more compact regenerator beds.
- Use of electrocalorific materials: (Northwest Food Processors Association)
- A new emerging cooling technology is based on the electrocaloric effect, whereby an electric field is cyclically applied to a paraelectric solid to cause heating and cooling. Analogous to magnetic refrigeration, which cyclically applies a magnetic field to a paramagnetic solid to cause heating and cooling (the magnetocaloric effect), a new technology is based on the electrocaloric effect, whereby an electric field is cyclically applied to a paraelectric solid to cause heating and cooling. The key difference between the two approaches is that large electrical fields are much easier and less expensive to produce than magnetic fields. If advantages can be confirmed through continued research and development, industry will once again have the opportunity to substantially reduce energy consumption by refrigerators and freezers.
- CeramPhysics, of Westerville, Ohio, has successfully replicated the work performed in Russia and is entering into a series of proprietary experiments designed to identify one or more alternate ceramic compositions with an electrocaloric effect larger than that of the composition used in the Russian research. The technology is in the conceptual stage of new product development.
- The energy-savings potential associated with this research is dramatic. According to the US Electric Power Research Institute, reducing the electrical consumption of US’s refrigerators and freezers by just 4.2 percent would save an estimated 479 million kWh—the equivalent of an average base-level power plant. At this rate, improving refrigerator and freezer energy efficiency by 50 percent would save 12 new base-level plants.
- Evaporator fan controls for refrigeration: (Northwest Food Processors Association,
Office of Industrial Technologies (2001), Summary of Program Results, U.S. Department of Energy, January 2001, www.oit.doe.gov, p.96)
- Evaporator fan controls for refrigeration: (Northwest Food Processors Association,
- In many facilities, refrigeration energy use can account for up to 40% of the total energy consumption. According to the Office of Industrial Technologies (OIT) (2001), a new technology, the Advanced Refrigeration Technologies (ART) Fan Controller, can reduce the costs of using these refrigeration units up to 50%.
- With assistance from the U.S. Department of Energy’s Inventions and Innovation Program, ART commercialized an innovative control strategy for walk-in refrigeration systems. The ART Evaporator Fan Controller is said to be inexpensive and easy to install (OIT 2001). It regulates the speed of the evaporator fan motors to meet the need of each phase of the refrigeration cycle. Just as energy is saved by turning off the lights in an unoccupied room, this controller saves energy by running the fans only as fast as the refrigerator needs at the time.
- According to the OIT (2001), in most cases ART customers will realize a 1- to 2-year return on investment by installing the controllers. For large corporate customers with hundreds of facilities, this can equate to a very large savings over a short period of time.
- Using a plate heat-exchanger for precooling ice-water with ammonia: (BAT in the Food, Drink and Milk Industries, June 2005)
- Ice-water is used as a cooling medium, e.g. for cooling milk and vegetables. The amount of energy consumed for the production of ice-water can be reduced by installing a plate heat-exchanger to precool the returning ice-water with ammonia, prior to a final cooling in an accumulating ice-water tank with a coil evaporator. This is based on the fact that the evaporation temperature of ammonia is higher in a plate cooler than when evaporator coils are used, i.e. -1.5°C instead of -11.5°C.
- It is reported that the capacity of an existing ice-water system can be increased without the need to increase the compressor capacity by installing a plate cooler for precooling the returning ice-water. In an example dairy, this precooling system saved almost 20% of electricity when installed in an existing ice-water system.
- b) Changes in the energy distribution system
No information is available.
- c) Changes in the heat supply system
No information is available.
Back to EFFICIENCY FINDER