Dyeing of flax
Back to EFFICIENCY FINDER FOR TEXTILE INDUSTRY
DYEING OF CELLULOSE FIBRES
1. DESCRIPTION OF TECHNIQUES, METHODS AND EQUIPMENT
Cellulose fibres can be dyed with a wide range of dyestuff, namely:
- reactive
- direct
- vat
- sulphur
- azoic (naphtol)
- Reactive dyes
One third of dyes used for cellulose fibres today are reactive dyes. They are mostly applied according to the pad-batch and continuous processes for woven fabric, while batch processes are the most common for knitted fabric, loose stock and yarn.
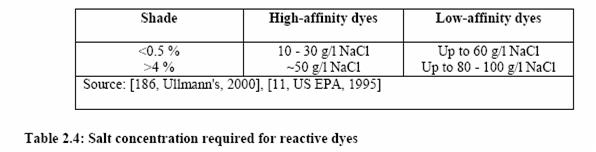
In batch dyeing, dye, alkali (sodium hydroxide or sodium carbonate or bicarbonate) and salt are added to the dye bath in one step, at the start of the process, or stepwise. In the stepwise process the alkali is added only after the dye has adsorbed to the fibre. Its amount is determined by the reactivity of the system and the desired depth of shade (cold dyers are applied at lower pH compared to warm and hot dyers). Salt is added to improve bath exhaustion: the concentration employed depends on the substantivity of the dye and on the intensity of the shade. Higher concentrations are required for deep shades and low-affinity dyes, as shown in the table below.
After dyeing, the liquor is drained off and the material is rinsed and then washed off with the addition of auxiliaries.
In pad dyeing processes dye and alkali can be added together to the dye liquor or in separate steps into two separate padders (or other types of application systems). When all the chemicals are applied in one step, the stability of the pad liquor is important. In fact with increasing reactivity of the dye there is a risk that the dye, after a long dwell time in the pad box, is hydrolysed by the alkali, before reacting with the fibre. For this reason dye and alkali are commonly metered separately into the padder. In addition, pad boxes are now constructed so that the liquor volume is as low as possible, so that it is replaced on average within 5 minutes [186, Ullmann’s, 2000].
Among semi-continuous processes the cold pad-batch is by far the most important one for reactive dyes. After the textile has been padded with dye and alkali, it is rolled up into batches. Fixing takes place during storage.
In continuous processes, padding, fixing, washing-off and drying are carried out in the same process line. Fixation is commonly achieved either by dry-heating or by steaming. The following processes are commonly used:
- pad-steam processes (one common method is the pad-dry-stream process which includes dye application by padding – intermediate drying – alkali application by padding – dye fixation with saturated steam – washing –drying)
- pad-dry thermofix processes (dye and alkali are padded at the same time; then the material can be dried and fixed in a single step or it can be thermo fixed after an intermediate drying stage)
In all cases, after fixation the material is always carefully washed off in open width or in a rope washing machine to remove completely the hydrolysed colourant and is then dried.
In pad-dry thermofix processes, urea is usually added to the padding liquor to act as a solvent for the dye during fixation. Urea melts at 115°C and binds water above 100°C. It can therefore be used as solvent for the dye in dry heat. A recently developed dyeing process is now available that does not require the addition of urea.
Urea is also sometimes used in pad-batch processes as dyeing solvent to increase the solubility of the dye. As early as 1992 the use of urea as dyeing solvent was already in decline [61, L. Bettens, 1999]. New highly soluble reactive dyes have been introduced in the market, which do not need urea even for deep dyeing in highly concentrated dye liquor.
- Direct dyes
Direct dyes are also quite important in cellulose fibres dyeing: 75 % of the total consumption of these colourants is used, in fact, to dye cotton or viscose substrates [186, Ullmann’s, 2000].
Direct dyes are applied directly from the dye bath together with salt (sodium chloride or sodium sulphate) and auxiliary agents, which ensure a thorough wetting and dispersing effect. Mixtures of non-ionic and anionic surfactants are used for this purpose.
In the batch process the dye is made into paste, then dissolved in hot water and added to the dye bath. The electrolyte is then added to the dye bath. After the dye bath has been drained, the fabric is washed with cold water and generally subjected to after-treatment.
Pad processes encompass the following techniques:
- pad-steam
- pad-roll
- cold pad-batch
- pad-jig process (he material is padded with the dye and then passed through a alt liquor in a jigger).
In all processes the material is rinsed at the end with cold water.
With increasing depth of colour the wt fastness can decrease to such an extent that after-treatment must generally be carried out [186, Ullmann’s, 2000]. Two methods exist:
- 1. removing the unfixed dye by washing with complexing agents or surfactants with a dispersing effect.
- 2. reducing the solubility of the dye by blocking the hydrophilic groups (“enlargement of the molecule”).
Various techniques can be applied to achieve this enlargementof the molecule. Namely, the dyed texile can be treated with:
- fixative cationic agents: these are complex substances that form with the anionic dye a salt-linek compound less soluble than the original dye. Quaternary ammonium compounds with long hydrocarbon chains, polyamines and polyethyleneimine derivates can be used for this purpose.
- metal salts: copper sulphate and potassium dichromate can form with certain azo dyes metal-complex with higher light fastness
- agents based on formaldehyde condenstation products with amines, polynuclear aromatic phenols, cyanamide or dicyanamide (the use of these condensation products leads to the formation of sparingly soluble adducts with the dye molecules)
- diazotised bases: after dyeing, the material is submitted to diazotisation and ir then coupled with aromatic amiens or pehols that must not contain hydrosolubilising groups [186, Ullmann’d, 2000].
Environmental concerns arise when after-treatment with formaldehyde condensation products or metal salts. The method using fixative cationic agents is, therefore, the most frequently applied. However, quaternary ammonium compounds are often non-biodegradable, fish-toxic and contain nitrogen.
- Vat dyes
Vat dyes have excellent fastness properties when properly selected and are often used for fabrics that will be subjected to severe washing and bleaching conditions (towelling, industrial and military uniforms etc.).
Vat dyes are normally insoluble in water, but they become water-soluble and substantive for the fibre after reduction in alkaline conditions (vatting). They are then converted again to the original insoluble form by oxidation and in this way they remain fixed into the fibre.
When applying vat dyes in batch processes the textile is dyed very rapidly and unevenly due to the high affinity of the dye. Nevertheless, level dyeing can be achieved by:
- adding levelling agents
- increase of the temperature under a controlled profile (“High temperature” process and “Semi-pigmentation” method)
- impregnation of the textile with the dye as water insoluble dispersion, followed by addition of the reductive agent in a subsequent step 8pre-pigmentation process)
In all cases, oxidation and after-treatment follow. After-treatment consists in washing the material in a weakly alkaline bath with a detergent at boiling temperature.
Continuous processes are used almost exclusively for dyeing woven fabrics and to the pad-steam process. The textile is padded with the aqueous dye dispersion in the presence of anti-migrant (polyacrylates, alginates, etc.) and dispersing/wetting agents, if required. After drying, the fabric is passed through a chemical padder, which contains the required amount of alkali and reducing agent and is fed immediately to a steamer. The material is finally rinsed, oxidised and soaped in an open-width washing machine.
Am more rapid, one-step process is also possible, but only for pastel to pale shades.
Voluminous open fabrics can be dyed according to a wet-steam process. Unlike the pad-steam process, this process does not require intermediate drying before steaming.
The following chemicals and auxiliaries are applied in vat dyeing:
- reducing agents: mainly sodium dithionite (hydrosulphite) and sulphoxylic acid derivates (Zn-sulphoxylate). The latter, in particular, is used when the pad-steam process is applied. Sulphur-free organic reducing agents such as hydroxyacetone are also now available for some applications
- oxidising agents, such as hydrogen peroxide, perborate, or 3-nitrobenzenesulphonic acid
- alkali (caustic soda)
- salt
- dispersing agents: they are already present in the dye formulation and are further added in the subsequent steps of the dyeing process
- levelling agents: they are already present in the dye formulation and are further added in the subsequent steps of the dyeing process
- levelling agents: they form adducts with the dye, thus retarding its absorption onto the fibre
- Sulphur dyes
Sulphur dyes are used in piece dyeing (cellulose and cellulose-polyester blends), yarn dyeing (sewing thread, warp yarn for denim fabric, yarn for coloured woven goods), dyeing of flock, card sliver (wool-man-made fibres blends) [186, Ullmann’s, 2000].
Like vat dyes, sulphur dyes are insoluble in water, and, under alkaline conditions, are converted into the leuco-form, which is water-soluble and has a high affinity for the fibre. After adsorption into the fibre the colourant is oxidised and conerted to the original insoluble state. The reducing agent, salts, alkali and unfixed dye are finally removed from the fibre by rinsing and washing.
Mostly continuous dyeing methods are applied, although batch dyeing (in jigger, jet and winch beck) is also possible.
In continuous processes the material is impregnated with dye, reducing agent and wetting agent through a one-bath or a two-bath procedure. With the one-bath procedure (pad-steam process) the reducing agent and the dye are added at the same time. With the two-bath procedure (pad-dry/pad-steam) the material is padded in the liquor containing the dye and the wetting agent, while the reducing agent is applied, if necessary, in a second step, after intermediate drying. The material is then submitted to air-free steaming. After that, rinsing, oxidation and re-rinsing are carried out. Because the exhaustion is not too high, it is possible to reuse dyeing baths in continuous processes.
Chemicals and auxiliaries applied to the substrate during the dyeing process are:
- reducing agents: sodium sulphide, sodium hydrogen sulphide and thiourea dioxide are the most commonly employed (although their use has decreased over the past decade [281, Belgium, 2002]). Binary systems made of glucose and sodium dithionite, hydroxyacetone and glucose are also used as alternative reducing agents.
- alkali (caustic soda)
- salt
- dispersing agents (they are necessary in the process steps in which the pigment has not yet been reduced or has been re-formed by oxidation)
- complexing agents: EDTA or polyphosphates are used in some cases, especially in circulating-liquor dyeing to avoid the negative effects of alkaline-earth ions on dyeing
- oxidising agents: mainly hydrogen peroxide and halogen-containing compounds such as bromate, iodate and chlorite.
- Azoic dyes (naphthol dyes)
Naphthol AS dyes allow colours with outstanding fastness, but their popularity has declined because of application costs and the complexity of the process for the preparation of the colourant. [77, EURATEX, 2000]
Dyeing with azoic colourants is a complex process which involves a number of delicate steps:
- preparation of the naphtholate solution by the hot solution process (the naphthol is dissolved by boiling with caustic soda) or by the cold solution process (the naphthol is solubilised with alcohol or cellosolve, caustic soda and cold water). For certain naphthols the addition of formaldehyde is also necessary to prevent the formation of free naphthol
- application of the naphtholate to the fibre by batch or padding techniques
- preparation of the diazotized base by reaction with sodium nitrite and hydrochloric acid (this step can be avoided when using fast colour salts)
- formation of the azoic dye into the fibre, by passing the textile, previously impregnated with the naphtholate solution, through a bath containing the diazotized base or the fast colour salt (addition of buffering agents is necessary to control pH, in order to increase the coupling capacity)
- after-treatment by rinsing the material to remove the excess naphthol from the fibre.
2. NEW TECHNOLOGIES
- a) Changes in the process
1.) Environmentally friendly method in reactive dyeing of cotton
The recipe for reactive dyeing of cotton can be divided into three steps: the pretreatment, the dye-bath and the final rinse. The consumption of chemicals and water in the rinse has been found to be considerable: three quarters of the total COD discharge of the recipe have relations to auxiliary chemicals used and three quarters of the total water consumption have relation to the rinse. The improvement potentials in the rinsing procedure were found to be considerable and the newly developed recipe focuses on improvements in the rinse. Tests with the new recipe have documented that neutralization, detergents and complexing agents can be left out completely with no adverse effect on product quality. All recognized tests of fastness and shade have been performed and evaluated in cooperation with the dyehouses. More than 50 full-scale dyeing experiments have been carried out in jets, overflow and drum batch machines. The experiments include 25 different reactive dyestuffs, among these azo. anthrachinon. phtalocyanin and formazan dyestuffs, including both monoreactive and bireactive dyestuffs of which 15 had the vinyl sulfone as reactive group. The dyestuffs have been combined in 20 different recipes covering very light to very dark shades. Hot membrane filtration of the hot rinsing water has been investigated in parallel projects and it is documented that membrane filtration saves chemicals, water and production time, and also gives large energy savings in one new recipe.
A chemical free, high temperature rinse, using a reduced number of batch rinses, is successfully documented. Neutralization before rinse and use of detergents and complexing agents during rinse can be left out completely. When introducing the new recipe, it is important that soft water - <3°dH - is used for the rinse. When omitting neutralization before rinsing in recipes using reactive dyestuffs with VS-reactive groups it is recommended to observe potential desorption of dyestuff carefully during the first rinse. The total water consumption can be reduced about 35-50% and the total time consumption for the recipe about 25%. In combination with hot membrane filtration of the hot rinsing water the reduction in water and time consumption are even higher and the energy consumption is reduced about 70%.
Source: “ENVIRONMENTALLY FRIENDLY METHOD IN REACTIVE DYEING OF COTTON”by Hans Henrik Knudsen and Henrik Wenzel; Institute for Product Development (IPU). Technical University of Denmark, Bygning 403ll, DK-2800 Lyngby, Denmark, 1996
- b) Changes in the heat supply system
No information is available.
- c) Changes in the energy distribution system
No information is available.