Difference between revisions of "Electroplating nickel in metal industry"
From Efficiency Finder
m (Protected "Electroplating nickel in metal industry" [edit=autoconfirmed:move=autoconfirmed]) |
|||
| Line 23: | Line 23: | ||
| − | [ | + | {| border="1" |
| + | | align="center" style="background:#2266CC;"|'''Process''' | ||
| + | | align="center" style="background:#2266CC;"|'''Temperature [°C]''' | ||
| + | | align="center" style="background:#2266CC;"|'''Heat transfer medium''' | ||
| + | | align="center" style="background:#2266CC;"|'''Residence time''' | ||
| + | | align="center" style="background:#2266CC;"|'''Chemicals''' | ||
| + | | align="center" style="background:#2266CC;"|'''Concentration''' | ||
| + | | align="center" style="background:#2266CC;"|'''Details''' | ||
| + | | align="center" style="background:#2266CC;"|'''Literature''' | ||
| + | |- | ||
| + | | style="background:#AACCEE;"| Nickel plating|| align="center"| 50-60|| align="center"| || align=”center”| || align=”center”| Nickel sulphate, nickel chloride and boric acid|| align="center"| Nickel sulphate: 240-357 g/l; nickel chloride: 35-60 g/l; boric acide: 30-45 g/l|| align="center"| Watts-type nickel solution|| rowspan="4" align="center"| BAT for the surface treatment of metals and plastics, August 2006 | ||
| + | |- | ||
| + | | style="background:#AACCEE;"| Plating with nickel sulphate-based solutions|| align="center"| 50-60|| align="center"| || align=”center”| || align=”center”| Nickel sulphamates (rather than sulphates), boric acid and frequently nickel chloride|| align="center"| Nickel sulphamate: 350-600 g/l of the tetrahydrate salt; boric acid: 35-45 g/l; nickel chloride: 1-15 g/l | ||
| + | |- | ||
| + | | rowspan="2" style="background:#AACCEE;"| Plating with nickel chloride- based solutions|| align="center"| 70|| align="center"| || align=”center”| || align=”center”| Nickel sulphamate (rather than sulphate), borica acid and frequently nickel chloride|| align="center"| || align="center"| for high concentrations: sulphamate solutions | ||
| + | |- | ||
| + | | | align="center"| 20-30|| align="center"| || align=”center”| || align=”center”| Nickel chloride hexahydrate, hydrochloric acid|| align="center"| Nickel chloride hexahydrate: 240 g/l; hydrochloric acid: 125 ml/g|| align="center"| Woods nickel strike solutions | ||
| + | |} | ||
| + | |||
Revision as of 10:12, 1 December 2010
Back to EFFICIENCY FINDER FOR METAL INDUSTRY
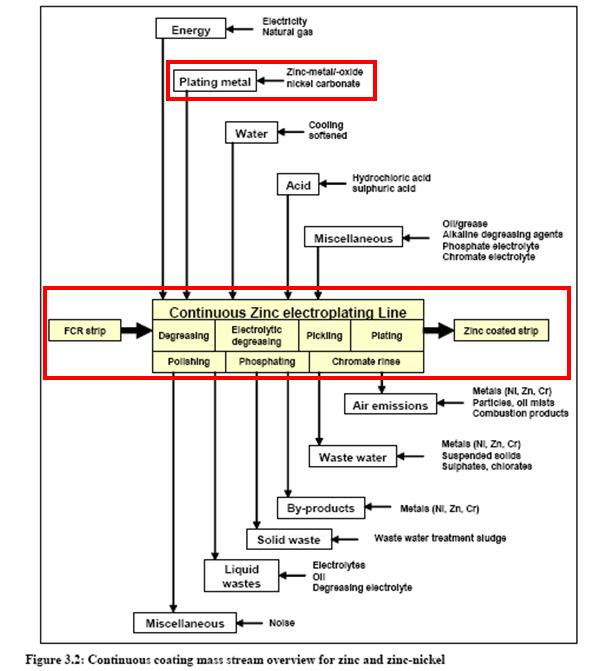
- Electroplating nickel flowsheet
Literature: BAT for the Surface Treatment of Metals & Plastics, 2006
Information about "Plant C" --> see below!
- Typical parameters of the process
| Process | Temperature [°C] | Heat transfer medium | Residence time | Chemicals | Concentration | Details | Literature |
| Nickel plating | 50-60 | Nickel sulphate, nickel chloride and boric acid | Nickel sulphate: 240-357 g/l; nickel chloride: 35-60 g/l; boric acide: 30-45 g/l | Watts-type nickel solution | BAT for the surface treatment of metals and plastics, August 2006 | ||
| Plating with nickel sulphate-based solutions | 50-60 | Nickel sulphamates (rather than sulphates), boric acid and frequently nickel chloride | Nickel sulphamate: 350-600 g/l of the tetrahydrate salt; boric acid: 35-45 g/l; nickel chloride: 1-15 g/l | ||||
| Plating with nickel chloride- based solutions | 70 | Nickel sulphamate (rather than sulphate), borica acid and frequently nickel chloride | for high concentrations: sulphamate solutions | ||||
| align="center"| 20-30 | Nickel chloride hexahydrate, hydrochloric acid | Nickel chloride hexahydrate: 240 g/l; hydrochloric acid: 125 ml/g | Woods nickel strike solutions |
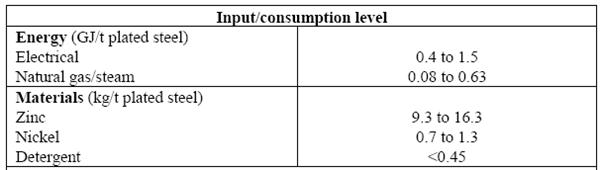
- Electroplating nickel
Literature: BAT for the Surface Treatment of Metals & Plastics, 2006
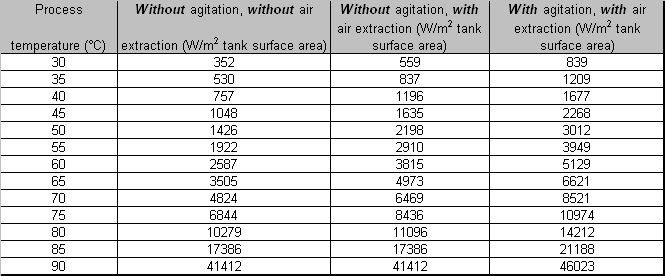
- Energy losses from the surface area of heated process solutions
LITERATURE: BAT for the Surface Treatment of Metals and Plastics, May 2005
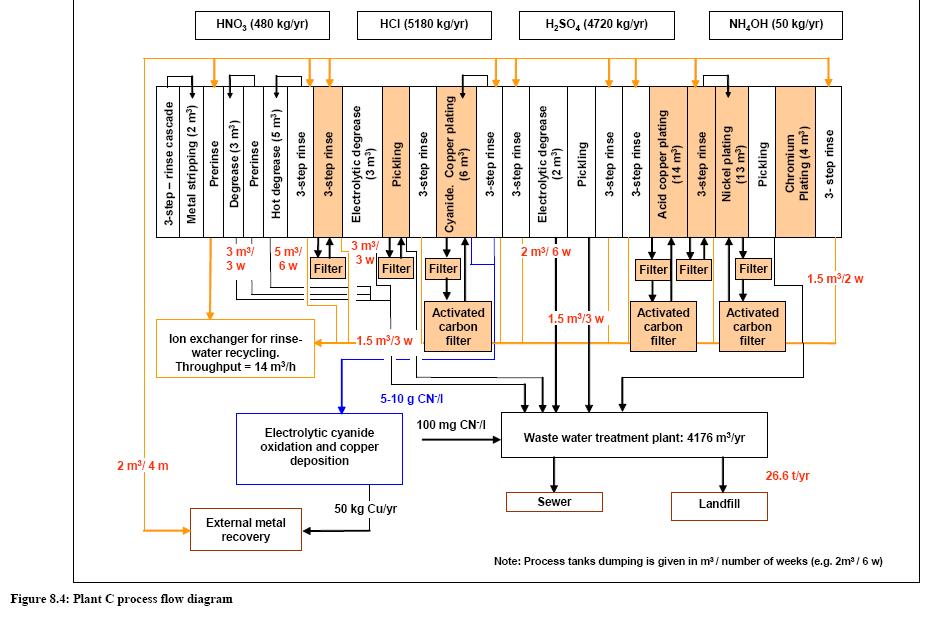
- Information about "plant C"
File:Information about "plant C".jpg